- Submit a Protocol
- Receive Our Alerts
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Phalloidin Staining of Actin Filaments for Visualization of Muscle Fibers in Caenorhabditis elegans
Published: Vol 11, Iss 19, Oct 5, 2021 DOI: 10.21769/BioProtoc.4183 Views: 3205
Reviewed by: Olli Matilainen Gawain McCollAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
E15.5 Mouse Embryo Micro-CT Using a Bruker Skyscan 1172 Micro-CT
Elena Astanina [...] Federico Bussolino
May 5, 2023 310 Views
THRIFTY—A High-throughput Single Muscle Fiber Typing Method Based on Immunofluorescence Detection
Sebastian Edman [...] William Apró
May 20, 2023 397 Views
Princeton RAtlas: A Common Coordinate Framework for Fully cleared, Whole Rattus norvegicus Brains
Emily Jane Dennis [...] Carlos D. Brody
Oct 20, 2023 567 Views
Abstract
Advances in C. elegans research have allowed scientists to recapitulate different human disorders, from neurodegenerative diseases to muscle dysfunction, in these nematodes. Concomitantly, the interest in visualizing organs affected by these conditions has grown, leading to the establishment of different antibody- and dye-based staining protocols to verify tissue morphology. In particular, the quality of muscle tissue has been largely used in nematodes as a readout for fitness and healthspan. Phalloidin derivatives, which are commonly used to stain actin filaments in cells and tissues, have been implemented in the context of C. elegans research for visualization of muscle fibers. However, the majority of the phalloidin-based protocols depend on fixation steps using harmful compounds, preparation of specific buffers, and large amounts of worms. Herein, we implemented a safer and more flexible experimental procedure to stain actin filaments in C. elegans using phalloidin-based dyes. Lyophilization of the worms followed by their acetone permeabilization allows bypassing the fixation process while also providing the opportunity to suspend the experiment at different steps. Moreover, by using conventional buffers throughout our protocol, we avoid the additional preparation of solutions. Finally, our protocol requires a limited number of worms, making it suitable for slow-growing C. elegans strains. Overall, this protocol provides an efficient, fast, and safer method to stain actin filaments and visualize muscle fibers in C. elegans.
Graphic abstract:
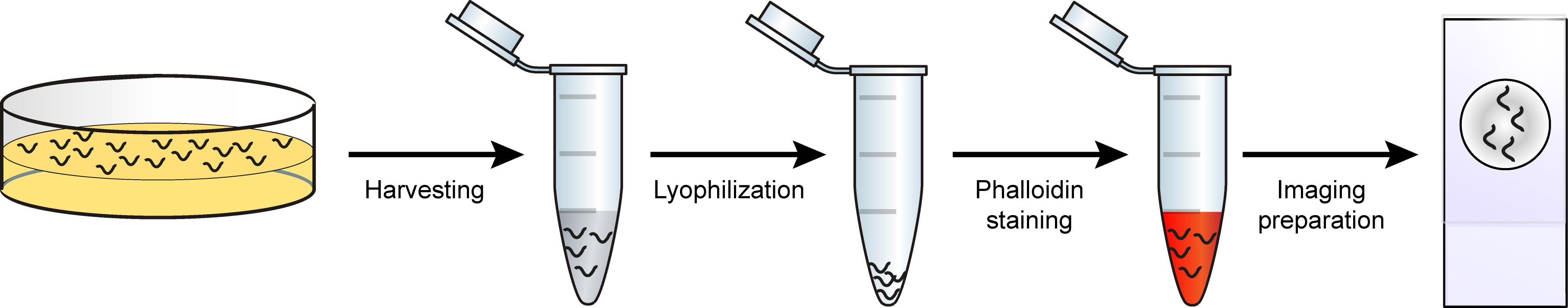
Schematic overview of phalloidin staining in C. elegans for assessing muscle fiber morphology.
Background
In the last decades, the nematode Caenorhabditis elegans has emerged as a powerful model organism and has provided an unprecedented opportunity to expand our knowledge in different fields of research, from developmental biology (Mello et al., 1992; Bowerman et al., 1993; Hubbard and Greenstein, 2000), to neuromuscular degeneration (Braungart et al., 2004; McColl et al., 2012; Sorrentino et al., 2017; Romani et al., 2021) and aging (Kimura et al., 1997; Herndon et al., 2002). One of the main reasons that promoted the usage of C. elegans as a laboratory model is that despite its simplicity, it has defined tissues. In particular, it consists of five main tissues: epidermis, reproductive tissues, digestive system, nervous system, and muscle tissue (Spencer et al., 2011). Among these, muscle tissue morphology and function have been widely used to typify C. elegans fitness and healthspan (Fire et al., 1991; Ryu et al., 2016; Fang et al., 2017; D’Amico et al., 2019; Romani et al., 2021). In recent years, numerous protocols have emerged to facilitate the visualization of C. elegans muscles, taking advantage of the transparent nature of this nematode. Phalloidin is a mushroom-derived molecule that specifically binds filamentous actin (F-actin); hence, phalloidin derivatives tagged with fluorescent molecules are commonly used in microscopy to visualize F-actin in different fields of research (Chazotte, 2010). Body wall muscles of the nematode C. elegans possess obliquely striated myofibrils composed of highly organized filaments of actin, whose disposition is altered in pathological conditions (Gieseler et al., 2018). For this reason, phalloidin-based staining of actin filaments has been successfully implemented in nematodes for the detection of cytoskeleton and muscle structure, with important applications in muscle disease and degeneration (Gieseler et al., 2018). However, phalloidin-based protocols typically rely on tedious fixation steps using harmful compounds, such as PFA, and require the preparation of specific, complicated buffers (Costa et al., 1997; Ono, 2001; Bansal et al., 2015; Geisler et al., 2020). We present here a method for C. elegans research that allows phalloidin staining of actin filaments, bypassing fixation, and using conventional buffers. Moreover, by integrating 4’,6-diamidino-2-phenylindole (DAPI) staining in our protocol, we allow the visualization of muscle cell nuclei, whose morphology can be used to interpret cellular homeostasis. Finally, our protocol requires a limited amount of worms and can be suspended at different steps, ensuring a versatile experimental procedure. Overall, our methodology provides a robust, simple, and safer pipeline for the visualization of muscle fibers and nuclei in C. elegans with applications in multiple fields of biomedical research. We predict that this protocol could be coupled with additional dyes specific for other tissues or cellular components, such as endoplasmic reticulum and mitochondria, allowing an even broader overview of worm physiology and homeostasis.
Materials and Reagents
1.5 ml Eppendorf tube (Eppendorf, catalog number: 11.3817.01)
Glass pipette
Microscope slides (Thermo Scientific, catalog number: 11950657)
Cover slips (VWR, catalog number: 43211.KG)
Plastic dropper (VWR, catalog number: AMP-30)
Aluminum foil
Liquid nitrogen
Acetone (Millipore, catalog number: 1.00014.1011)
Rhodamine Phalloidin (Invitrogen, catalog number: 219920-04-4)
Methanol (Sigma-Aldrich, catalog number: 646377-1L)
BSA (Sigma-Aldrich, catalog number: A7906-100G)
Tween-20 (Sigma-Aldrich, catalog number: P1379)
DAPI (Invitrogen, catalog number: D1306)
Agarose (Promega, catalog number: V3125)
M9 buffer (see Recipes)
Washing buffer (see Recipes)
2 µg/ml DAPI in M9 (see Recipes)
2% (w/v) agarose (see Recipes)
Equipment
Standard equipment for worm culture (see Brenner, 1974)
Centrifuge (Eppendorf, Centrifuge 5424)
SpeedVac Vacuum Concentrator (Labogene, Speed Scan 40)
Laboratory fume hood
Leica SP8 confocal microscope (Stand: Upright Leica DM6 CS; Illumination: Lumencor Sola SM II LED, Laser; Software: LAS-X; Camera: DFC 7000 GT (B/W); Objective: HC PL APO, 63×/1.40 oil, DIC POL0.14)
Software
Fiji (Schindelin et al., 2012)
Procedure
Culture 50 worms on a NGM agar plate. Each plate of 50 worms serves as a replicate
Note: The protocol will work with any number of worms between 50 and 100. We suggest using two replicates for each experimental condition.
Worm retrieval from agar plates
Wash worms off the plate using 1 ml of M9 buffer and place the solution in a 1.5 ml Eppendorf tube.
Note: We suggest retrieving the M9 solution from the plates using a glass pipette, which reduces the adhesion of worms to the pipette’s walls.
Spin briefly in a centrifuge to pellet the worms but not the bacteria (do not exceed 400 × g); 10 s is sufficient to pellet adult worms.
Remove as much supernatant as possible using a micro-pipette (1,000 µl and 200 µl).
Note: C. elegans tend to swim, so the pellet is not stable; avoid bringing the tip of the pipette too close to the pellet to prevent sucking in any worms.
Repeat steps from 1 to 3 one more time to ensure that all the worms are retrieved from the plate.
Snap freeze the worms by immerging the Eppendorf tube in liquid nitrogen.
Note: At this stage, worms can be stored at -80°C for months.
Worm permeabilization
Open the Eppendorf tube and place it in a SpeedVac Vacuum Concentrator to dry the worm pellet at 20°C. This step typically takes ~10 min but can vary according to the amount of supernatant that is left from Step B3.
Add three drops of ice-cold acetone to the dry worms. Gently tap the Eppendorf tube five times.
Note: It is important to cover all the worms; additional drops of acetone could be used if necessary. If worms are not correctly permeabilized, the protocol will fail.
Incubate 5 min at room temperature.
Remove as much of the acetone as possible using a micro-pipette (1,000 µl and 200 µl) and air dry the remaining acetone in a laboratory fume hood.
After complete evaporation of the acetone, worms can be stored at -20°C for months.
Phalloidin staining
Note: We suggest working in a dark environment to prevent phalloidin degradation and consequent failure of the protocol.
Place 2.5 U (for each replicate) of Rhodamine Phalloidin in an Eppendorf tube.
Use the SpeedVac Vacuum Concentrator to remove the methanol in which the Rhodamine Phalloidin is dissolved.
Note: It takes ~5-10 min to dry the Rhodamine Phalloidin, but this varies according to the volume that is used.
Resuspend the dried Rhodamine Phalloidin in 20 µl (for each replicate) of M9.
Add 20 µl of the M9-resuspended Rhodamine Phalloidin to the dried worms. Gently tap the Eppendorf tube five times.
Incubate in the dark at room temperature for 30 min.
Wash the worms twice in 1 ml of washing buffer.
If nuclei staining is not required, proceed to Procedure F.
DAPI staining
Note: We suggest working in a dark environment to prevent DAPI degradation.
Resuspend the worms in 20 µl of 2 µg/ml DAPI in M9. Gently tap the Eppendorf tube five times.
Incubate in the dark at room temperature for 5 min.
Wash the worms twice in 1 ml of washing buffer.
Preparation of the worms for imaging
Note: We suggest working in a dark environment to prevent degradation of the fluorescent dyes and consequent failure of the protocol.
Resuspend the worms in 10 µl of M9.
Prepare a 2% (w/v) agarose pad on a microscope slide for each replicate. To prepare the agarose pad, apply lab tape on two microscope slides and place a third one in between them (Figure 1a). Apply 2-3 drops of agarose in the center of the microscope slide not wrapped in tape using a micro-pipette (1,000 µl). Place another microscope slide on the top of the agarose (Figure 1b). Wait 1-2 min for the agarose to solidify and remove the microscope slide on the top. Store the agarose pads in a humid place before usage to prevent dryness.
Note: One layer of tape is sufficient to create the space needed for the formation of an agarose pad. Cut the micro-pipette tip to facilitate the dispensation of agarose. Gently put the last microscope slide on the liquid agarose to avoid the formation of bubbles in the agarose pad.

Figure 1. Agarose pads preparation. First (a) and second (b) steps for the preparation of agarose pads described in Step F2.Transfer all the M9 containing the worms to the agarose pad using a glass pipette.
Apply a cover slip, avoiding the formation of bubbles. Surface tension of M9 will keep the cover slip steady during the imaging process but avoid touching it.
Imaging using a confocal microscope
Note: We suggest not using inverted microscopes to keep the cover slip steady during the imaging process.
Images analysis
Use Fiji to open the images recorded with the confocal microscope.
Add scale bar: “Analyse” -> “Tools” -> “Scale bar” (Figure 2).
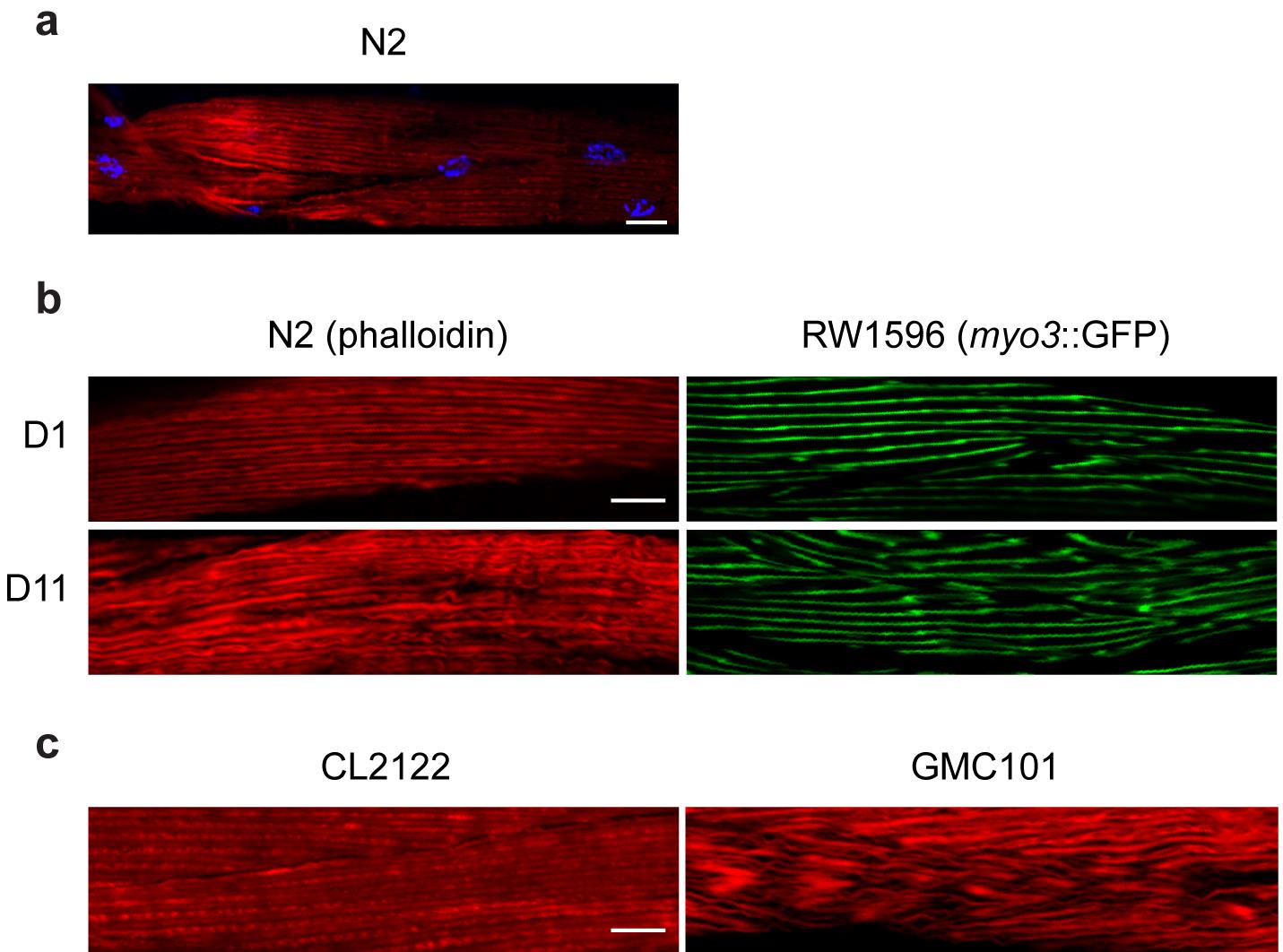
Figure 2. Phalloidin staining applications and comparison with GFP-tagged muscle fibers. (a) Representative image of day 1 adult N2 worms; in red, actin filaments stained with Rhodamine-Phalloidin; in blue, nuclei stained with DAPI. (b) Day 11 adult N2 worms present disorganized muscle fibers, visualized here through Rhodamine-Phalloidin staining (in red), when compared to young (day 1) worms as described before (Romani et al., 2021). These results are comparable to those obtained using a transgenic worm strain (RW1596) in which the muscle fibers are labeled with GFP (in green). (c) Rhodamine-Phalloidin staining of the inducible muscle proteotoxicity model, GMC101, reveals altered muscle fiber morphology compared to the control strain CL2122, consistent with the data obtained in young and old worms and as described before (McColl et al., 2012). Scale bar, 10 µm; magnification, 63×.
Recipes
NGM agar plates (see Brenner, 1974)
M9 buffer (Table 1)
Table 1. M9 buffer composition. List of components and volumes for 1 L of working stock.Components Quantity Na2HPO4 6 g KH2PO4 3 g NaCl 5 g MgSO4∙7H2O 250 mg Water 1 L Note: Filter before using.
Washing buffer
M9 1 ml
BSA 50 mg
Tween-20 50 μl
2 µg/ml DAPI in M9
To make a 5 mg/ml DAPI stock solution, dissolve the content of one vial (10 mg) in 2 ml of water. For long-term storage the stock solution can be aliquoted and stored at ≤ -20°C. For short-term storage the solution can be kept at 2-6°C, protected from light.
2% (w/v) agarose
Dissolve 500 mg of agarose in 25 ml of water.
Note: Warm up the solution to near-boiling temperature to dissolve the agarose. We suggest using a microwave for this step.
Acknowledgments
The research in the J.A. lab was supported by the EPFL, the European Research Council (ERC-AdG-787702), the Swiss National Science Foundation (SNSF 310030B_160318 and 31003A_179435), the National Research Foundation of Korea (GRL 2017K1A1A2013124), the Fondation Suisse de Recherche sur les Maladies Musculaires (FSRMM), and the Fondation Marcel Levaillant. D’Amico et al. (2019), Romani et al. (2021), and Sorrentino et al. (2017) used this procedure for staining and visualizing muscle fibers in C. elegans.
Competing interests
The authors have no competing interests to declare.
References
- Bansal, A., Zhu, L. J., Yen, K. and Tissenbaum, H. A. (2015). Uncoupling lifespan and healthspan in Caenorhabditis elegans longevity mutants. Proc Natl Acad Sci U S A 112(3): E277-286.
- Bowerman, B., Draper, B. W., Mello, C. C. and Priess, J. R. (1993). The maternal gene skn-1 encodes a protein that is distributed unequally in early C. elegans embryos. Cell 74(3): 443-452.
- Braungart, E., Gerlach, M., Riederer, P., Baumeister, R. and Hoener, M. C. (2004). Caenorhabditis elegans MPP+ model of Parkinson’s disease for high-throughput drug screenings. Neurodegener Dis 1(4-5): 175-183.
- Brenner, S. (1974). The Genetics of Caenorhabditis elegans. Genetics 77(1): 71-94.
- Chazotte, B. (2010). Labeling cytoskeletal F-actin with rhodamine phalloidin or fluorescein phalloidin for imaging. Cold Spring Harb Protoc 2010(5):pdb.prot4947.
- Costa, M., Draper, B. W. and Priess, J. R. (1997). The role of actin filaments in patterning the Caenorhabditis elegans cuticle. Dev Biol 184(2): 373-384.
- D’Amico, D., Mottis, A., Potenza, F., Sorrentino, V., Li, H., Romani, M., Lemos, V., Schoonjans, K., Zamboni, N. and Knott, G. (2019). The RNA-Binding Protein PUM2 Impairs Mitochondrial Dynamics and Mitophagy During Aging. Mol Cell 73(4): 775-787.e10.
- Fang, E. F., Waltz, T. B., Kassahun, H., Lu, Q., Kerr, J. S., Morevati, M., Fivenson, E. M., Wollman, B. N., Marosi, K. and Wilson, M. A. (2017). Tomatidine enhances lifespan and healthspan in C. elegans through mitophagy induction via the SKN-1/Nrf2 pathway. Sci Rep 7: 46208.
- Fire, A., Albertson, D., Harrison, S. W. and Moerman, D. G. (1991). Production of antisense RNA leads to effective and specific inhibition of gene expression in C. elegans muscle. Development 113(2): 503-514.
- Geisler, F., Coch, R. A., Richardson, C., Goldberg, M., Bevilacqua, C., Prevedel, R. and Leube, R. E. (2020). Intestinal intermediate filament polypeptides in C. elegans: Common and isotype-specific contributions to intestinal ultrastructure and function. Sci Rep 10: 3142.
- Gieseler, K., Qadota, H. and Benian, G. M. (2018). Development, structure, and maintenance of C. elegans body wall muscle. WormBook.
- Herndon, L. A., Schmeissner, P. J., Dudaronek, J. M., Brown, P. A., Listner, K. M., Sakano, Y., Paupard, M. C., Hall, D. H. and Driscoll, M. (2002). Stochastic and genetic factors influence tissue-specific decline in ageing C. elegans. Nature 419: 808-814.
- Hubbard, E. J. and Greenstein, D. (2000). The Caenorhabditis elegans gonad: a test tube for cell and developmental biology. Dev Dyn 218(2): 2-22.
- Kimura, K. D., Tissenbaum, H. A., Liu, Y. and Ruvkun, G. (1997). daf-2, an insulin receptor-like gene that regulates longevity and diapause in Caenorhabditis elegans. Science 277(5328): 942-946.
- McColl, G., Roberts, B. R., Pukala, T. L., Kenche, V. B., Roberts, C. M., Link, C. D., Ryan, T. M., Masters, C. L., Barnham, K. J. and Bush, A. I. (2012). Utility of an improved model of amyloid-beta (Aβ1-42) toxicity in Caenorhabditis elegans for drug screening for Alzheimer’s disease. Mol Neurodegener 7: 57.
- Mello, C. C., Draper, B. W., Krause, M., Weintraub, H. and Priess, J. R. (1992). The pie-1 and mex-1 genes and maternal control of blastomere identity in early C. elegans embryos. Cell 70(1): 163-176.
- Ono, S. (2001). The Caenorhabditis elegans unc-78 Gene Encodes a Homologue of Actin-Interacting Protein 1 Required for Organized Assembly of Muscle Actin Filaments. J Cell Biol 152(6): 1313-1320.
- Romani, M., Sorrentino, V., Oh, C. M., Li, H., de Lima, T. I., Zhang, H., Shong, M. and Auwerx, J. (2021). NAD+ boosting reduces age-associated amyloidosis and restores mitochondrial homeostasis in muscle. Cell Rep 34(3): 108660.
- Ryu, D., Mouchiroud, L., Andreux, P. A., Katsyuba, E., Moullan, N., Nicolet-Dit-Félix, A. A., Williams, E. G., Jha, P., Lo Sasso, G. and Huzard, D. (2016). Urolithin A induces mitophagy and prolongs lifespan in C. elegans and increases muscle function in rodents. Nat Med 22(8): 879-888.
- Schindelin, J., Arganda-Carreras, I., Frise, E., Kaynig, V., Longair, M., Pietzsch, T., Preibisch, S., Rueden, C., Saalfeld, S. and Schmid, B. (2012). Fiji: an open-source platform for biological-image analysis. Nat Methods 9: 676-682.
- Sorrentino, V., Romani, M., Mouchiroud, L., Beck, J. S., Zhang, H., D’Amico, D., Moullan, N., Potenza, F., Schmid, A. W. and Rietsch, S. (2017). Enhancing mitochondrial proteostasis reduces amyloid-β proteotoxicity. Nature 552(7684): 187-193.
- Spencer, W. C., Zeller, G., Watson, J. D., Henz, S. R., Watkins, K. L., McWhirter, R. D., Petersen, S., Sreedharan, V. T., Widmer, C. and Jo, J. (2011). A spatial and temporal map of C. elegans gene expression. Genome Res 21: 325-341.
Article Information
Publication history
Accepted: Jul 5, 2021
Published: Oct 5, 2021
Copyright
© 2021 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Romani, M. and Auwerx, J. (2021). Phalloidin Staining of Actin Filaments for Visualization of Muscle Fibers in Caenorhabditis elegans . Bio-protocol 11(19): e4183. DOI: 10.21769/BioProtoc.4183.
Category
Developmental Biology > Morphogenesis > Cell structure
Developmental Biology > Cell growth and fate > Myofiber
Cell Biology > Tissue analysis > Tissue imaging
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Tips for asking effective questions
+ Description
Write a detailed description. Include all information that will help others answer your question including experimental processes, conditions, and relevant images.
Share
Bluesky
X
Copy link